Better fat bubbles could power a new generation of mRNA vaccines
New lipid delivery systems aim to improve potency and reduce side effects

Medical officer prepares a dose of Moderna COVID-19 vaccine during the third dose inoculation program for health workers in Mangusada Hospital, Bali, Indonesia on August 4 2021. Indonesian health workers receives the third dose COVID-19 vaccine using mRNA based vaccine from Moderna in order to boost their body’s immune system and protect them from the new variant of coronavirus which has surged over the past few weeks in Indonesia. (Photo by Johanes Christo/NurPhoto via Getty Images)
JOHANES CHRISTO/NURPHOTO
As any dietician will tell you, some fats are good—and that is surely true of the little fatty balls found in two of the world’s most widely used COVID-19 vaccines. Known as lipid nanoparticles (LNPs), these tiny bubbles of fat encase messenger RNA (mRNA) that encodes a viral protein, helping ferry it into cells and shield it from destructive enzymes. The technology was key to the success of COVID-19 shots from Moderna and the Pfizer-BioNTech collaboration. But as beneficial as these fats are, there is plenty of room for improvement.
The nanoparticles are a major source of unwanted side effects when they spread through the body, triggering the aches and inflammation many people experience after vaccination. They do a poor job of unloading their cargo once inside cells, a necessary step for the proteinmaking machinery to turn the mRNA sequences into immune-priming signals. And because they tend to fall apart when warm, they have to be stored at low temperatures, limiting their global use.
“This is a system that clearly has legs,” says biochemist Pieter Cullis of the University of British Columbia (UBC), Vancouver, who created the first LNPs, but “we still need to increase the efficiency of LNPs—that’s for sure.”
A new generation of LNPs with greater potency, fewer side effects, increased stability, and more precise tissue-targeting properties is now under development at big pharma and biotech startups. Big money is at stake: These improved nanoparticles could lead to better mRNA vaccines for COVID-19 and other diseases. They might also help mRNA deliver on its promise as a therapeutic tool to treat disease. “There are innovations on delivery that certainly could change the game,” says Philip Santangelo, a biomedical engineer at the Georgia Institute of Technology who has collaborated with several mRNA companies.
Cullis and colleagues developed the first LNPs about 20 years ago to carry gene-silencing drugs into cells. He and others later tailored the LNPs’ four lipid components to deliver disease-correcting mRNA to faulty cells. Now that they are being put to a new use, in vaccines, “There is still so much optimization and development that needs to happen,” says UBC bioengineer Anna Blakney, co-founder of the RNA vaccine company VaxEquity. And when it comes to understanding how cells interact with the nanoparticles, “it’s just this big question mark,” she adds.
One clue emerged earlier this year when Genentech scientists showed how nanoparticles activate a particular inflammatory pathway, the interleukin-1 axis, which is critical to generating protective immune responses but can also spur side effects. Among the LNPs tested, one made with SM-102, an “ionizable” lipid that helps bind and package mRNA into LNPs, proved an especially strong instigator of this pathway. That could help explain why Moderna’s shot, which relies on SM-102, is both highly effective and prone to making people feel icky.
The Genentech team did not evaluate the comparable lipid found in the Pfizer-BioNtech vaccine. But Mohamad-Gabriel Alameh and colleagues from the University of Pennsylvania Perelman School of Medicine tested a closely related one and found that it triggered a wide range of inflammatory molecules, both desired and not. The goal now is to design ionizable lipids that activate favorable immune pathways without overstimulating detrimental ones, says Alameh, who co-founded AexeRNA Therapeutics with bioengineer Michael Buschmann of George Mason University and others. “Is it very simple? No,” Alameh says, but it should be possible.
Before his death in March, Buschmann led a team that in 2021 showed how the electric charge of LNPs is critical to vaccine success. A negative charge makes the particle less likely to stay in the muscle and lymph nodes of injected mice, where it could elicit beneficial immune responses; instead it tends to spread widely, raising the risk of fevers, chills, and other adverse reactions.
To make a less negatively charged nanoparticle, the researchers tweaked the chemistry of the ionizable lipid. When formulated into an mRNA vaccine for COVID-19, the new LNP carrier prompted mice to produce more protective antibodies than standard delivery systems and had fewer side effects, according to data posted last year in preprint form and now under review at Nature Communications.
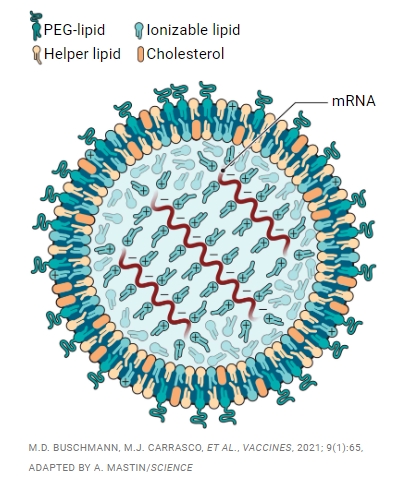
The particulars of nanoparticles
To boost vaccine potency and limit side effects, researchers are altering each of the four ingredients that make up lipid nanoparticles. Each particle includes ionizable lipids that bind messenger RNA (mRNA) and shift their charge from positive to neutral once in the body to limit the particle’s toxicity. The three other types of fats contribute to its structure and stability. Helper lipids also aid particles in fusing with cells, cholesterol helps them escape from cells’ endosomes, and polyethylene glycol (PEG)-lipids prevent them from clumping to help prolong their action.
Dan Peer, a biochemist at Tel Aviv University and co-founder of the vaccine delivery startup NeoVac, has also developed libraries of new ionizable lipids with atypical structures. In unpublished experiments, they seem to enable better mRNA vaccines with fewer side effects and also extend their shelf stability at room temperatures.
Other improvements could come from boosting the uptake of LNPs into cells and then enhancing their ability to break free of the sacs of cell membrane, known as endosomes, that carry them inside. The vast majority of LNPs get trapped in these receptacles and then destroyed or ejected without delivering their vaccine payloads, meaning “there’s a huge amount of RNA that’s not being used,” says Gaurav Sahay, a bioengineer at Oregon Health & Science University.
The shape of ionizable lipids affects an LNP’s ability to disrupt an endosome, as does cholesterol, one of the other fats in LNPs. Together with Moderna scientists, Sahay and colleagues reported in 2020 that using different forms of cholesterol can enhance rates of LNP escape from endosomal entrapment. He has founded a company called Enterx Biosciences to commercialize his discoveries.
Sanofi has begun to evaluate some of its customized LNPs head-to-head in human trials. In a study launched in 2021, for example, the company assessed two LNP options for delivering an mRNA flu shot it is developing. According to preliminary data, one lipid formulation proved much better at kick-starting anti-influenza immunity, Frank DeRosa, head of research and biomarkers at Sanofi’s mRNA Center of Excellence, announced at an investor event in December 2021. But the same LNP also provoked more frequent side effects at higher doses.
Other firms, including BioNTech and Arcturus Therapeutics, have begun to explore ways to eliminate polyethylene glycol, a compound that helps stabilize LNPs but has also been linked to some types of bad vaccine reactions. Many more companies, meanwhile, are focused on optimizing lipids for delivering mRNA to treat disease rather than prevent it. This requires getting mRNAs that encode disease-correcting proteins to the precise cells and tissues where they are needed—not just to the liver, where current LNP formulations tend to end up following infusion. “Delivery of LNPs will be key to really expanding the reach of mRNA” beyond preventative vaccines, says Dominik Witzigmann, co-founder and chief executive of the Cullis-founded startup NanoVation Therapeutics.
The heightened focus on LNP technologies, along with the profits reaped from the COVID-19 vaccines, has brought increased litigation. Alnylam, which helped develop the first approved medicine delivered in an LNP—a gene-silencing drug marketed since 2018 to treat a rare neurodegenerative disorder—claims that its foundational patents cover lipid components of the Moderna and Pfizer-BioNTech vaccines. And Arbutus BioPharma, yet another Canadian firm co-founded by Cullis, is seeking damages from Moderna for allegedly infringing on a patent that covers LNPs comprising certain ratios of lipids.
But these intellectual property disputes are unlikely to have a chilling effect on LNP innovation, says Jacob Sherkow, a biotech patent attorney with the University of Illinois College of Law. “There’s too much money at play.”
